Background On Microcosm Testing
MICROCOSM TEST
FOR NATURAL ATTENUATION OF CHLORINATED SOLVENTS
Margaret Findlay, Ph.D. and Samuel Fogel, Ph.D.
Bioremediation Consulting Inc.
39 Clarendon Street, Watertown MA 02472
Introduction
Microcosm testing procedures have been recommended by the US AF and US EPA as a method for documenting natural attenuation of chlorinated solvents in ground water and evaluating a site’s potential for in situ remediation by enhanced natural attenuation. Microcosms are small samples of soil and ground water which are maintained as microbial habitats under conditions simulating those of the sample’s original location. Samples from anaerobic areas thought to support microbial degradation of chlorinated solvents by the process of reductive dechlorination must be manipulated and maintained under conditions which exclude oxygen. The demonstration of contaminant dechlorination within a closed microcosm provides significant evidence of on-going natural attenuation. Due to the high degree of control that the microcosm offers for confining all of the products of a series of dechlorination reactions, and therefore of obtaining a mass balance for the proposed decontamination process, microcosm test results are in some ways more conclusive than those of field monitoring. We describe simplified microcosm preparation and maintenance, as well as methods for monitoring ongoing microcosm tests for changes in key chemical and microbiological parameters.
Anaerobic Transformations of PCE and TCE
Tetrachloroethylene (PCE) and trichloroethelyene (TCE) in ground water can be biodegraded by anaerobic bacteria using a biochemical process known as reductive dechlorination. Electrons are removed from low molecular weight organic compounds (the electron donor or food source) and biochemcally used to replace a chlorine atom of the contaminant, e.g. PCE. In this case, PCE acts as the electron acceptor.
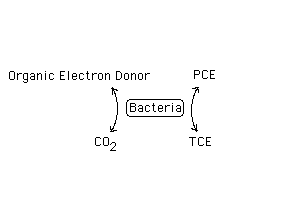
If ground water conditions are optimal, and sufficient energy is available from organic electron donors, this process can proceed until all of the chlorine atoms are removed, and PCE or TCE is dechlorinated completely via cis-dichloroethene (cDCE) and chloroethene (VC) to ethene gas, a harmless end-product.
PCE > TCE > cDCE > VC > Ethene
When PCE is transformed to ethene by ground water bacteria, without human intervention, the process is termed natural attenuation. At most sites, reductive dechlorination is incomplete due to insufficient electron donor. When electron donor compounds are purposely added to overcome this limitation, the process is termed enhanced natural attenuation. Both natural attenuation and enhanced natural attenuation can be demonstrated using laboratory microcosms.
Several Microbial Types are Involved in Reductive Dechlorination.
While the removal of the chlorine atom is carried out only by special bacteria called dechlorinators, participation of the entire subsurface anaerobic microbial community is important in supporting this process. Facultative aerobes and nitrate reducers initially lower the redox potential by consuming organic compounds while reducing oxygen to water and nitrate to ammonia or N2 . Fermenters break down complex sugars of plant origin to volatile fatty acids such as formate, acetate, propionate and butyrate. The fermenters as well as acetogens produce hydrogen, which may be the immediate source of electrons used by the dechlorinators. Sulfate reducers, also obtaining electrons from fatty acids and other low molecular weight compounds, reduce sulfate to HS` , lowering the redox potential to the -300 mv level required by the dechlorinators. The non-dechlorinators also produce nutritional factors such as vitamins, which may be required by the dechlorinators. The only pure culture capable of dechlorinating PCE to ethene requires added vitamin B12 and H2 (Maymo-Gatell, 1995).
Microcosm Tests are a Valuable Supplement to Field Measurements.
At sites containing PCE or TCE contamination, the existence of daughter products DCE, VC and ethene give the first indication that natural attenuation is occurring or has occurred either at the sampling site or up-gradient. At many PCE sites, dechlorination occurs very slowly because the ground water lacks the necessary bacterial food sources and mineral nutrients. As a consequence, annual or semiannual samples from the same well yield imprecise data relative to documenting small concentration changes, so that years of sampling are required before convincing evidence of on-going natural attenuation is obtained. In a series of wells in the plume, aligned along the direction of ground water flow, a downgradient increase in the ratio of daughter products to parent compounds indicates past or on-going natural biodegradation. Microcosm testing, in which dechlorination is observed in an isolated sample of ground water and soil in a tightly sealed bottle, provides the most convincing evidence of on-going attenuation because the data allows calculation of a mass balance for the dechlorination, in which the molar increase in breakdown products is equal to the molar amount of initial chlorinated contaminant.
Two Types of Microcosm Tests.
The EPA Method. The US EPA (Wiedemeier et al.,1998) recommends a design for microcosm tests to be carried out for the purpose of estimating the in situ rate constant for the biodegradation of contaminant at the sample location. For this purpose, the method requires that microcosm bottles contain a high ratio of solids to ground water, closely simulating subsurface conditions, and that the bottles be completely filled, leaving minimal or no headspace. These tests require sacrifice of entire microcosms for extraction and analysis at each time point, and are cumbersome and expensive. Because natural attenuation is often very slow, these tests can require 12 months to complete.
The Air Force Method. Simplified microcosm test procedures have been recommended by the Air Force (Morse et al, 1998) which are not designed to determine the rate constant for natural attenuation, but rather to determine whether natural attenuation is occurring at a site, and to evaluate the potential for enhancing the natural attenuation by testing amendments which would speed the natural process. (A similar test was described by Findlay et al., 1995). The simplified tests use microcosms containing a soil-ground water slurry with 60 cc (or more) headspace. The large headspace allows removal, at several time intervals, of small gas samples for (less costly) direct analysis of volatile contaminant and daughter product concentrations, avoiding the requirement of sacrificing test bottles at each time interval. Because electron donor is added to stimulate the natural process in some of the microcosm bottles, useful results can usually be obtained in 3 to 6 months.
Microcosm Set-up.
The procedures described here are those used at BCI Lab, and are generally in accordance with those recommended by Morse et al 1998, (RABITT Protocol). Field ground water and soil samples are collected and transported using methods which exclude oxygen. In the laboratory, transfer of the samples into microcosm bottles is conducted in a glovebox having a non-oxygen atmosphere. The microcosm bottle, which allows oxygen to be excluded and an anaerobic environment to be maintained for bacteria (Figure 1), is a 160 ml glass ‘serum’ bottle having a narrow neck and small opening. After introducing the sample, the bottle is sealed with a Teflon-lined rubber septum which is tightly affixed with a crimped aluminum cap. The septum excludes oxygen, prevents the escape of the organic solvents, and the rubber portion re-seals punctures made by the narrow gauge needles used to remove small samples for analysis during the test.
For the simplified microcosm tests, the bottles are not completely filled with the soil-ground water mixture but have a 60 cc headspace into which a fraction of each volatile organic compound moves, in accordance with its Henry’s Law constant. Although the headspace method does not exactly mimic subsurface conditions, it allows the progress of the dechlorination to be monitored by removing a 0.1 cc of headspace with a syringe, and directly injecting this sample into a gas chromatograph. Careful selection of GC column and temperature program allows the separation and quantitation of several chlorinated contaminants (such as PCE, 1,1,1-TCA, freon, carbon tetrachloride, and their dechlorination products, including ethene and ethane).
If contaminant concentration is very low, the microcosms can be spiked with PCE or TCE in order to obtain measurable concentrations of transformation products. Killed control bottles are made by autoclaving or adding strong acid, in order to assess abiotic conversions. To evaluate the potential for enhanced natural attenuation, additional microcosms are set up and amended with electron donors (such as lactic acid) and minerals.
Analytical Methods used by BCI Lab to Measure Microcosm Activity.
BCI monitors microbial processes during microcosm tests by removing 0.1 cc headspace or aqueous samples and analyzing these directly according to the methods listed in Table 1. At the initiation of a microcosm test, if sugar has been added as electron donor, fermentors, if present, break down the sugar, producing organic acids and H2, which are analyzed by capillary ion electrophoresis and reduction gas analyzer, respectively.
If NO3 is present, nitrate reducers convert NO3 to N2 or NH3, and if SO4 is present, sulfate reducers convert SO4 to H2S. Acetogens may convert other organic acids to acetate, producing additional H2. The depletion of the organic acid supply by these organisms is monitored by electrophoresis, and additional organic acids are then added to the microcosm if needed. Phosphate, a mineral nutrient required by bacteria, is also monitored by electrophoresis, and replenished as necessary.
During the sulfate reduction phase, PCE dechlorination to TCE and subsequently to DCE may occur, and is monitored by direct injection of headspace samples into the gas chromatograph. As H2 builds up in the microcosm, we anticipate the initiation of DCE dechlorination to VC and ethene, and continue monthly monitoring by gas chromatography. During the final stages of ethene production, the ethene may be further reduced to ethane, which is also monitored by gas chromatography. Methane may accumulate and appear in the gas chromatography analysis, as methanogens produce methane utilizing H2 and CO2, or acetate.
|
Table 1. Analytical Methods Used by BCI Lab for Monitoring Microcosms |
||
|
Anaerobic Bacteria |
Activity |
Analytical Method |
|
Fermentors |
Convert sugar to organic acids and H2 |
Capillary Ion Electrophoresis(1) Reduction Gas Analyzer(2) |
|
Sulfate Reducers |
Reduce sulfate produce H2S convert PCE to DCE |
C.I. Electrophoresis chemical test kit. Gas Chromatography(3) |
|
Acetogens |
produce acetic acid produce H2 |
C.I. Electrophoresis Reduction Gas Analyzer |
|
Methanogens |
produce CH4 |
Gas Chromatography |
|
Dechlorinators |
convert PCE to ethene |
Gas Chromatography |
(1) Waters Corporation (2) Trace Analytical (3) Hewlett Packard
Case Study: TCE Dechlorination in Samples from Chemical Manufacturing Site.
Figures 2A and 2B show the results from a microcosm study using ground water and sediment from a monitoring well at a chemical manufacturing site. The microcosms initially contained about 2 ppm TCE, 1 ppm cDCE and 0.3 ppm VC. Headspace samples were removed at intervals and analyzed for each chlorinated contaminant as well as for the dechlorination product, ethene.
Dechlorination using native electron donor. The ground water from this site initially contained about 35 ppm acetate (0.6 mM) which may have accumulated as a result of microbial metabolism of non-chlorinated contaminants such as methanol and dichloromethane. These microcosms showed conversion of TCE to DCE and VC by day 60. Starting on day 30, conversion of VC to ethene was initiated, with the complete dechlorination process requiring 90 days. Figure 2A summarizes the data by plotting the decrease in the combined molar concentration of chlorinated compounds, with the concomitant increase in the molar ethene concentration. A mass balance evaluation. can easily be made from this figure, which shows that all of the biodegraded chlorinated ethenes are recovered as ethene.
Dechlorination using native electron donor plus organic acid amendment. In addition to the acetate already present in the ground water, these microcosms received 0.43 mM each of formic, acetic, lactic and butyric acids as supplementary electron donors. Dechlorination of the contaminants was more rapid in these microcosms. TCE was completely converted to cDCE in 15 days, and DCE was converted to VC by day 30. Dechlorination of VC to ethene was initiated on day 20 and was complete by day 60. Again, a good mass balance was obtained, with recovered ethene accounting for all lost chlorinated compounds (Figure 2B).
Other Monitoring Wells. Ground water from other wells in the plume did not contain native electron donor. The microcosms constructed from these wells showed dechlorination only when amended with electron donor, indicating that site remediation would benefit from injection of electron donor compounds in these areas.
Application of Microcosm Testing for Field Remedial Design.
Microcosm testing can provide information for the design of field pilot tests and full-scale remediations including selection of site specific donor compounds and mineral nutrients, decisions regarding bioaugmentation, and design of remedial strategy.
Selection of site specific donors: The microbes at a site may be acclimated to the use of specific electron donors that have been present at the site as co-contaminants. By comparing the rate of dechlorination in microcosms amended with different donors, the preferred electron donor for a given site can be determined.
Evaluation of mineral nutrient requirement. Lack of nitrogen and phosphorus compounds, which are required for microbial growth, may limit natural attenuation or may become limiting if enhancement with electron donor induces microbial growth. Comparing the rate of dechlorination in microcosms with and without added phosphate and ammonia can indicate whether site ground water should be amended with these compounds.
Bioaugmentation: In cases where microcosm testing indicates that reductive dechlorination does not respond to amendments with electron donor, biaoaugmentation (adding bacteria) can be evaluated by inoculating microcosms with ground water from other site locations. By identifying those portions of the contaminated site which contain dechlorinators, ground water recirculation can be designed to assure distribution of the desired microbes.
Anaerobic / Aerobic two zone treatment: At those sites where incomplete conversion of PCE has occurred and DCE and VC have accumulated, two-zone anaerobic/aerobic treatment may be a cost-effective option which can be investigated by microcosm testing. This approach would make use of methane-oxidizing bacteria, which are present in many anaerobic ground waters, and can readily co-metabolically oxidize DCE and VC to CO2 . To make use of these bacteria, a downgradient zone would be aerated, allowing the methane oxidizers to grow using methane previously produced in the upgradient anaerobic zone (Fogel et. al., 1995).
Conclusion.
The microcosm test procedures, utilizing headspace analysis of volatile contaminants and breakdown products, can be useful in documenting natural attenuation, evaluating the potential of enhanced attenuation, selecting amendments, and planning field remedial strategy.
References
Findlay, M., A. Leonard, S. Fogel and W. Mitchell. 1995. "Laboratory Biodegradation to Define In Situ Potential for PCE Transformations". Third International In Situ and On-Site Bioreclamation Symposium: In Situ Potential for PCE Transformation. In R. Hinchee, C. Vogel, F. Brockman (Eds.), Microbial Processes for Bioremediation, 3(8) 223-229., Battelle Press, Columbus, Ohio.
Fogel, S., R. Lewis, D. Groher and M. Findlay. 1995. "PCE Treatment in Saturated Soil Columns: Methanogens in Sequence with Methanotrophs". Third International In Situ and On-Site Bioreclamation Symposium: In R.E. Hinchee, A. Leeson, L. Semprini (Eds.), Bioremediation of Chlorinated Solvents 3(4) pp. 153-160. Battelle Press, Columbus, Ohio.
Maymo-Gatell, X., V. Tandoi, J. Gossett, and S. H. Zinder. 1995. Characterization of an H2-utilizing anaerobic enrichment culture that reductively dechlorinates tetrachloroethene to vinyl chloride and ethene in the complete absence of methanogenesis and acetogenesis. Appl. Environ. Micorbiol. 61: 3928-3933.
Morse, J., B. Alleman, J. Gossett, S. Zinder, D. Fennell, G. Sewell, C. Vogel. 1998. Draft Technical Protocol: A Treatability Test for Evaluating the Potential Applicability of the Reductive Anaerobic Biological In Situ Treatment Technology (RABITT) to Remediate Chloroethenes. (Appendix C.3.4. Design, implementation, and interpretation of microcosm studies)
Wiedemeier, T., M. Swanson, D. Mouton, E. Gordon, W. Wilson, B. Wilson, D. Campbell, P. Haas, R. Miller, J. Hansen, F. Chapelle. 1998. Technical Protocol for Evaluating Natural Attenuation of Chlorinated Solvents in Ground Water. EPA/600/R-98/128/ Office of Research and Development Washington DC 20460.
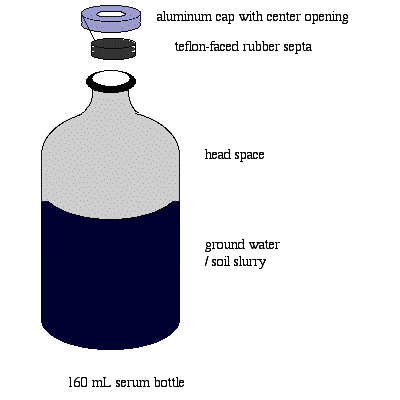
Figure 1. Microcosm Container, consisting of 160 ml serum bottle, showing Teflon-faced rubber septa ready to be affixed by crimping the aluminum cap around the rim of the bottle. For anaerobic studies, the headspace is flushed with oxygen-free gas during set-up.
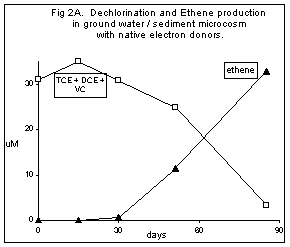
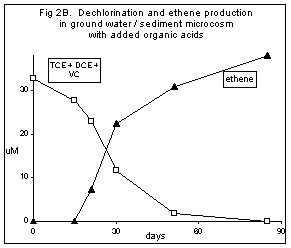
Figure 2. Microcosm test results for anaerobic incubation of ground water and sediment from a monitoring well at a chemical manufacturing site. Data obtained by gas chromatography of 0.1 cc headspace samples removed by syringe, expressed as micromolar concentrations in the aqueous portion. Open squares are the sum of the TCE, cDCE, and VC. Figure 2A, native electron donor only. Figure 2B, native electron donor plus amendment with additional organic acids as electron donors.
Acknowledgement
This article was written for publication in the Journal Soil Sediment & Groundwater February/ March 2000.